

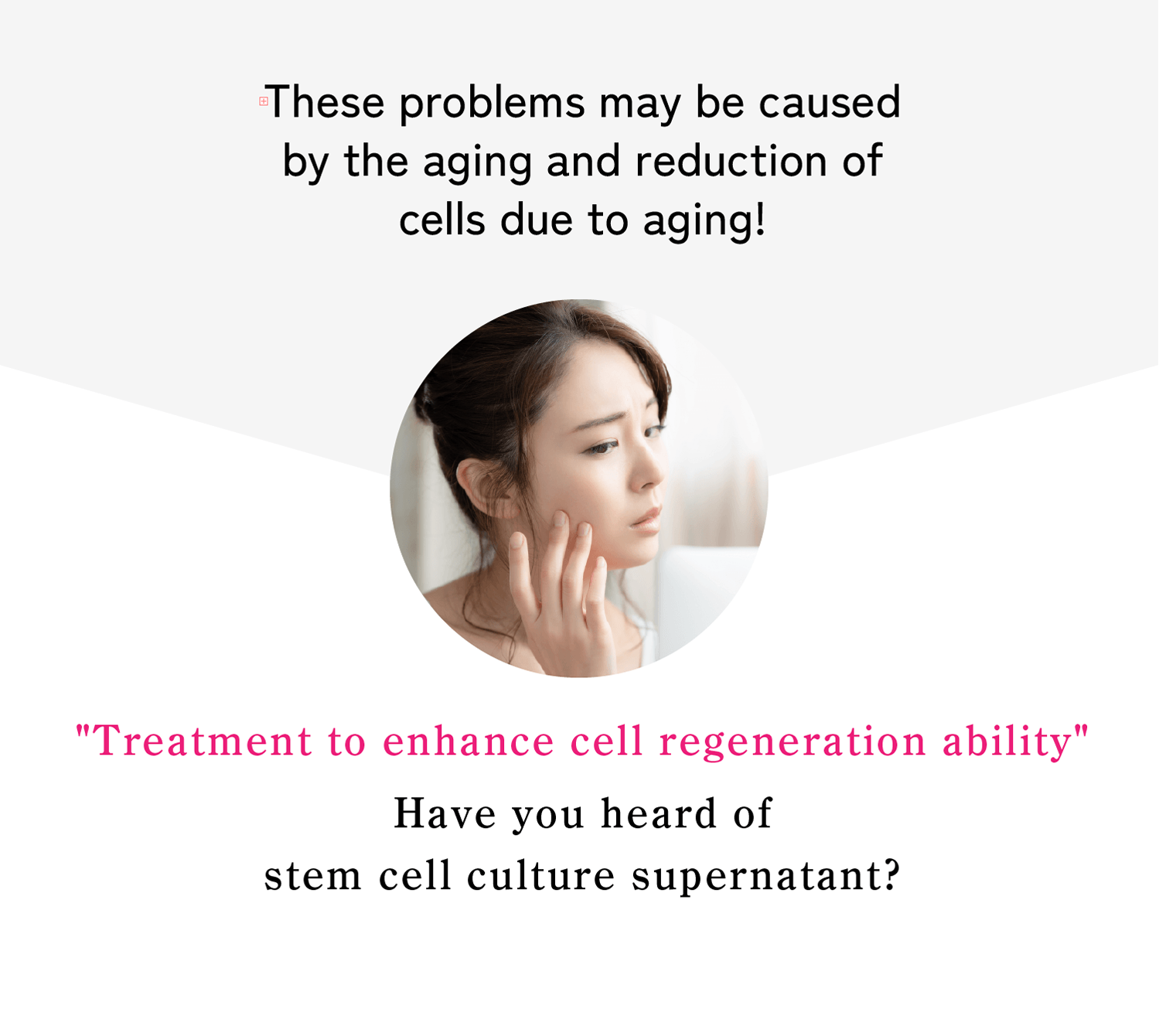
Recommended for individuals with the following concern
- I want to regain my youthful appearance.
- I want to achieve clearer, brighter skin.
- I want to reduce wrinkles and age spots.
- I want to restore firmness and elasticity to my skin.
- I want to improve dark circles under my eyes.
- I want to treat acne and visible pores.
- I want to address thinning hair and hair loss.
- I have trouble concentrating.
- I want to relieve joint pain.
Recommended for those with the following concerns
- I want to regain my youthful appearance.
- I want to achieve clearer, brighter skin.
- I want to reduce wrinkles and age spots.
- I want to restore firmness and elasticity to my skin.
- I want to improve dark circles under my eyes.
- I want to treat acne and visible pores.
- I want to address thinning hair and hair loss.
- I have trouble concentrating.
- I want to relieve joint pain.


Enhances cellular function and stimulates regeneration
The supernatant is a fluid obtained by centrifuging the culture medium used to grow human-derived stem cells, then sterilizing it.
Stem cells have the remarkable ability to differentiate into various types of tissue and organs, which is why stem cell-based regenerative medicine has gained significant attention.
Additionally, stem cell culture supernatant can revitalize cells weakened by aging and other factors, and is therefore expected to offer a wide range of beauty benefits.
This is because the supernatant contains cytokines (growth factors) essential for anti-aging.

Cytokines are powerful substances that promote cell proliferation and differentiation. Stem cell culture supernatant contains cytokines (growth factors), which are signaling molecules secreted by stem cells.
Rich in cell-activating signaling compounds, the supernatant helps restore cells weakened by aging and other factors. Cytokines support the repair of damaged tissues and help reactivate cell function throughout the body.
Acidic Fibroblast Growth Factor
Stimulates and promotes the proliferation of dermal stem cells (fibroblasts), leading to wound healing, wrinkle reduction, and skin brightening effects.
Epidermal Growth Factor
It signals epidermal stem cells to increase the production of epidermal cells, promoting skin turnover (metabolism). This helps improve blemishes and dullness.
Insulin-Like Growth Factor
It supports the regeneration of new skin cells and stimulates the production of collagen, elastin, and hyaluronic acid, helping to restore firmness and elasticity to the skin.
Hepatocyte Growth Factor
It plays a key role in tissue regeneration by promoting cell proliferation and motility not only in the liver, but also in skin cells, while also inducing morphogenesis and angiogenesis.
Transforming Growth Factor-β
It has anti-inflammatory properties and helps soothe and improve sensitive skin.
Platelet-Derived Growth Factor
It stimulates and promotes the division of stem cells involved in tissue repair, including fibroblasts, adipose-derived stem cells, and epithelial stem cells.
Vascular Endothelial Growth Factor
It plays a vital role in angiogenesis—the formation of new blood vessels through the branching and extension of existing ones.
Keratinocyte Growth Factor
It stimulates hair matrix stem cells, promoting hair growth and increased thickness.
Basic Fibroblast Growth Factor
It acts on vascular, skin, muscle, and other cell types, regulating cell differentiation and proliferation. It also plays a role in the formation of nerves and bones.
Stem cell culture supernatant, rich in cytokines, helps restore cells damaged by aging and other factors, offering a range of benefits for both health and beauty.

Feature 1
Safety Assessment
Impurities are thoroughly removed to ensure maximum safety.
Feature 1
Safety Assessment
Impurities are thoroughly removed to ensure maximum safety.
A groundbreaking technology that redefines stem cell culture supernatant!
In the medical community, it was not widely known that cells release harmful substances such as ammonia, much like the human body does.
As a result, conventional stem cell culture supernatant products on the market were not properly refined, with impurities making up approximately 80% of the content.
However, our purified and concentrated stem cell culture supernatant—produced using a newly developed purification method—has successfully removed these impurities to the greatest extent, achieving the highest quality in the world.
Manufactured in Japan using 100% human-derived components
All ingredients in our purified and concentrated stem cell culture supernatant are 100% human-derived, with no animal-derived components used in the manufacturing process.
The stem cells are sourced exclusively from healthy Japanese female donors, and the product is manufactured entirely in Japan, ensuring both safety and quality you can trust.
Extensively screened to ensure the absence of viruses
| Hepatitis B Virus (HBV) |
| Hepatitis C Virus (HCV) |
| Human Immunodeficiency Virus (HIV) |
| Human T-Cell Leukemia Virus (HTLV) |
| Parvovirus B19 (PVB-19) |
The purified and concentrated stem cell culture supernatant is completely free of infectious viruses, bacteria, and DNA fragments.
Safety is ensured through rigorous screening of Japanese female donors and comprehensive testing for harmful substances and viruses in the final product prior to shipment.
Feature 2 Quality Evaluation Exceptional Cytokine Concentration
Feature 2
Quality Evaluation
Exceptional Cytokine Concentration
One of the most important growth factors found in stem cell culture supernatant is HGF (Hepatocyte Growth Factor).
HGF promotes the formation of new blood vessels and supports tissue repair.
It also helps break down fibrous tissue, such as collagen, which often accumulates in damaged or necrotic tissue.
The concentration of other active ingredients is said to be proportional to the amount of HGF present.
Until now, commercially available culture supernatants have shown significant variations in composition between batches—even within the same product line.
We have resolved this issue by successfully controlling and standardizing the content, ensuring consistent quality in every lot.


This is a medical treatment that aims to regenerate damaged or lost tissues in the human body due to illness, injury, or aging.
In clinical practice, stem cell-based therapies have become the mainstream approach in regenerative medicine.
In recent years, regenerative medicine has attracted significant attention as a groundbreaking treatment method and is now being applied to the field of aesthetic medicine.
What Are Stem Cells?
Stem cells play a vital role in the regeneration of human tissue. Simply put, they are unique cells capable of replenishing and repairing damaged or lost tissues.
Unlike ordinary cells, which can only replicate themselves—blood cells become blood, and skin cells become skin—stem cells possess the ability to develop into various types of cells.
As normal cells age or become damaged, their regenerative capacity declines.
However, stem cells are versatile cells that can develop into a wide range of cell and organ types.
They can regenerate and repair tissues that ordinary cells cannot, offering possibilities beyond the scope of conventional treatments.
That’s why our clinic offers safe regenerative medicine by harvesting a very small amount of the patient’s own stem cells, culturing them, and re-administering them.
This approach minimizes the risk of immune rejection and complications.

Why Are Stem Cells So Effective?
When damage occurs in the body, the affected area sends out a signal requesting repair. Stem cells detect this signal, migrate to the site of injury, and transform into the necessary cell types to repair and regenerate tissue, helping to improve symptoms.
It is believed that the strength of this biological signal is one reason why stem cell therapy tends to be more effective in patients during the acute phase of illness or injury, compared to those in the chronic phase.
Rather than removing the affected tissue or replacing it with artificial materials, the goal is to rebuild it using the patient’s own cells. There have been numerous reports of symptom improvement in cases previously considered difficult or irreversible with conventional treatments.

Shibuya Cosmetic Surgery Clinic
is officially certified with a Type 2 Regenerative Medicine Provision Plan Number.
Our clinic has been officially approved by the Ministry of Health, Labour and Welfare to provide Type 2 regenerative medicine.
Stem cell-based regenerative medicine is subject to rigorous evaluation by a Special Certified Regenerative Medicine Committee, authorized by the Ministry of Health, Labour and Welfare. This review assesses the treatment's appropriateness, safety, physician qualifications, and cell processing management system.
If the committee deems the plan appropriate, it can be submitted to the Ministry of Health, Labour and Welfare for approval, after which treatment may begin.
Shibuya Cosmetic Surgery Clinic is a certified medical facility that has completed this formal process and obtained a Type 2 Regenerative Medicine Provision Plan Number.
■ About the Regenerative Medicine Provided
Type 2 Regenerative Medicine
Plan Number: PB3210106
"Treatment for chronic pain using autologous adipose-derived stem cells"
[Affiliated Research Institution]
Cell culture is outsourced to Cell Bank Co., Ltd. (Facility Number: FA3150017).



Shibuya Cosmetic Surgery Clinic
is officially certified with a Type 2 Regenerative Medicine Provision Plan Number.
Our clinic has been officially approved by the Ministry of Health, Labour and Welfare to provide Type 2 regenerative medicine.
Stem cell-based regenerative medicine is subject to rigorous evaluation by a Special Certified Regenerative Medicine Committee, authorized by the Ministry of Health, Labour and Welfare. This review assesses the treatment's appropriateness, safety, physician qualifications, and cell processing management system.
If the committee deems the plan appropriate, it can be submitted to the Ministry of Health, Labour and Welfare for approval, after which treatment may begin.
Shibuya Cosmetic Surgery Clinic is a certified medical facility that has completed this formal process and obtained a Type 2 Regenerative Medicine Provision Plan Number.
■ About the Regenerative Medicine Provided
Type 2 Regenerative Medicine
Plan Number: PB3210106
"Treatment for chronic pain using autologous adipose-derived stem cells"
[Affiliated Research Institution]
Cell culture is outsourced to Cell Bank Co., Ltd. (Facility Number: FA3150017).




A world-renowned professor of plastic surgery at Jichi Medical University,who is also an expert in regenerative medicine, serves as our technical advisor.

A world-renowned professor of plastic surgery at Jichi Medical University,who is also an expert in regenerative medicine, serves as our technical advisor.

Over 22 years since opening, with more than 400,000 procedures performed.
Our doctor is a board-certified plastic surgeon recognized by the Japan Society of Plastic Surgery.
*From February 1999 to November 2021

Over 22 years since opening, with more than 400,000 procedures performed.
Our doctor is a board-certified plastic surgeon recognized by the Japan Society of Plastic Surgery.
*From February 1999 to November 2021

Repeat rate: over 90%.We take the time to carefully create a personalized treatment plan that best suits your individual needs.

Repeat rate: over 90%.We take the time to carefully create a personalized treatment plan that best suits your individual needs.

I have extensive experience in cosmetic medicine,with many years of practice as a cosmetic dermatologist specializing in laser treatments for pigmentation removal, hyaluronic acid injections, Botox, thread lifts, and a wide range of skin rejuvenation and anti-aging procedures.

I have extensive experience in cosmetic medicine,with many years of practice as a cosmetic dermatologist specializing in laser treatments for pigmentation removal, hyaluronic acid injections, Botox, thread lifts, and a wide range of skin rejuvenation and anti-aging procedures.


The stem cell culture supernatant is administered via injection into the arm.
Rich in cytokines, the supernatant is expected to activate the patient’s own stem cells, promoting tissue regeneration and repair.
For cosmetic purposes, it may help rejuvenate the skin and improve overall skin appearance.

Stem Cell Culture Supernatant
| First Trial | 99,000 yen |
| Single Session | 165,000 yen |
| 10 Sessions | 990,000 yen |
Risks and Side Effects
Potential adverse reactions may include internal bleeding, localized pain or swelling, chills, nausea, fever, skin redness, or rash.

- How is it administered?
-
It is administered via injection into the arm.
- Is it painful?
-
You may feel a slight sting when the needle is inserted, similar to a standard injection.
- Are there any side effects?
-
Some individuals may experience allergic reactions. If you have any concerns, please consult with your doctor in advance.
- How many times do I have to take it to see results?
-
We generally recommend one to two sessions per month.
The appropriate frequency and number of sessions will be tailored to your individual condition. Please consult with your doctor during your initial visit for a personalized plan.
Medical Equipment and Medications Used
- This product has not been approved under Japan’s Pharmaceuticals and Medical Devices Act.
- The unapproved drugs used in this treatment have not received approval under the Pharmaceuticals and Medical Devices Act.
They are legally provided through in-house dispensing (partially outsourced). In Japan, the use of unapproved drugs is permitted under the prescribing physician’s responsibility. - There are no other approved drugs in Japan with the same ingredients or properties.
- Potential serious side effects may not yet be fully known.
![[Official] For cosmetic surgery and cosmetic surgery, Shibuya Cosmetic Surgery Clinic](/images/logo_img.png)













